 This way Wilhelm Ostwald has Kelvin-Planck Statement explained.įor the operation of the heat engine, a continuous exchange of heat has to take place. Furthermore, Wilhelm Ostwald introduced this device and it would perform work by absorbing energy from a body as heat. Hence, experts refer to work as high-grade energy, and heat as low-grade energy.Ĭonsider a hypothetical device known as Perpetual motion machine of the second kind (PMMSK). However, heat’s conversion cannot take place fully into work. Here, the full conversion of the work can take place into heat. 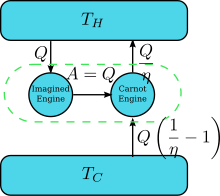
This means that the conversion of work can take place into heat and vice-versa. Furthermore, both these forms are inter-convertible and both follow the S.I unit Joules. Most noteworthy, clubbing together both these statements gave birth to the Kelvin-Planck Statement.Īn important point to remember is that heat (Q) and Work (W) happen to be the two forms of energy. Planck’s statement tells us that, for the reversible system, the total sum of entropies would remain constant. 
Kelvin Planck Statement Understanding Kelvin Planck StatementĪccording to Kelvin statement, it is not possible by cooling below the highest cooling temperature of the surrounding objects to derive mechanical effect from any matter. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
